This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison
What is Proteomics?
Proteomics is the study of proteins. One way to study proteins is to look at post-translational modifications, or protein modifications. Phosphorylation is one form of modification that can occur, and is most often seen on the amino acids: serines, threonines, and tyrosines. The benefit to phosphorylation is the versatility and reversibility of the modifications. With different combinations of amino acid phosphorylation, chemically unique proteins can be formed. These modifications are important in regulating many different pathways, and in diseased states phosphorylation can be disturbed. In these diseased states, proteins can no longer function properly, which can be due to improper folding or protein-protein interactions that are due to mis-phosphorylation. [1]
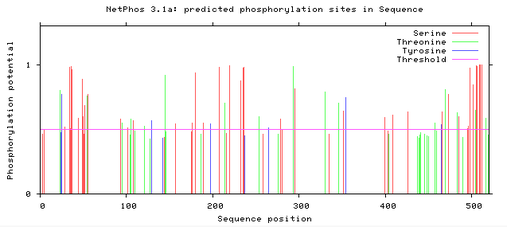
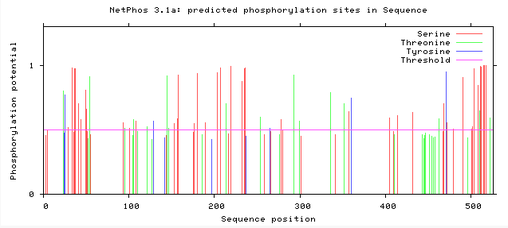
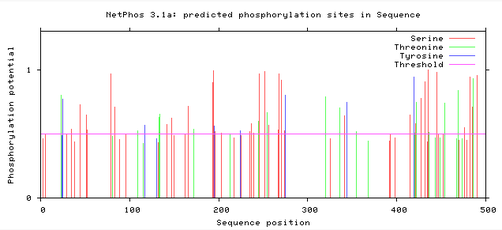
MITF protein phosphorylation sites
Below are readouts from NetPhos 3.1, that show the predicted phosphorylation sites in the MITF protein for Humans, Mice, and Zebrafish. The horizontal red line shows the threshold for potential phosphorylation sites, and anything that reaches above the line is significantly phosphorylated within a cell. There are three differently colored lines which depict the different amino acids: Red – Serine, Green – Threonine, and Blue – Tyrosine. [2]
Analysis
The phosphorylation potential sites are all very similar across humans, mice, and zebrafish. In all three there are sites free of phosphorylation around the sequence position 80 and 300-400. These sites both correlate with the domains of the MITF protein. At sequence position 300 is the binding domain HLH, which suggests that for proper MITF protein function this domain needs to be relatively free of phosphorylation.
References
1. Hunter, T. (2012). Why nature chose phosphate to modify proteins. Philosophical Transactions of the Royal Society B: Biological Sciences,367 (1602), 2513-2516. doi:10.1098/rstb.2012.0013
2. NetPhos 3.1 Server. (n.d.). Retrieved March 22, 2017, from http://www.cbs.dtu.dk/services/NetPhos/
1. Hunter, T. (2012). Why nature chose phosphate to modify proteins. Philosophical Transactions of the Royal Society B: Biological Sciences,367 (1602), 2513-2516. doi:10.1098/rstb.2012.0013
2. NetPhos 3.1 Server. (n.d.). Retrieved March 22, 2017, from http://www.cbs.dtu.dk/services/NetPhos/